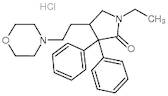
CAS: 113-07-5 - Doxapram Hydrochloride
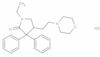
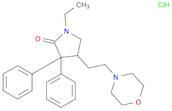
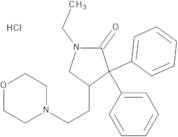
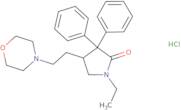
Formel:C24H33ClN2O3
InChl:InChI=1S/C24H30N2O2.ClH/c1-2-26-19-22(13-14-25-15-17-28-18-16-25)24(23(26)27,20-9-5-3-6-10-20)21-11-7-4-8-12-21;/h3-12,22H,2,13-19H2,1H3;1H
InChI Key:InChIKey=MBGXILHMHYLZJT-UHFFFAOYSA-N
SMILES:Cl.O=C1N(CC)CC(CCN2CCOCC2)C1(C=3C=CC=CC3)C=4C=CC=CC4
- Synonyme:
- 1-Ethyl-4-(2-Morpholinoethyl)-3,3-Diphenylpyrrolidin-2-One Monohydrochloride
- 1-Ethyl-4-(2-morpholinoethyl)-3,3-diphenyl-2-pyrrolidinone hydrochloride
- 1-Ethyl-4-[2-(Morpholin-4-Yl)Ethyl]-3,3-Diphenylpyrrolidin-2-One Hydrochloride (1:1)
- 2-Pyrrolidinone, 1-ethyl-4-(2-(4-morpholinyl)ethyl)-3,3-diphenyl-, monohydrochloride
- 2-Pyrrolidinone, 1-ethyl-4-(2-morpholinoethyl)-3,3-diphenyl-, hydrochloride
- 2-Pyrrolidinone, 1-ethyl-4-(2-morpholinoethyl)-3,3-diphenyl-, monohydrochloride
- 2-Pyrrolidinone, 1-ethyl-4-[2-(4-morpholinyl)ethyl]-3,3-diphenyl-, hydrochloride (1:1)
- 3,3-Diphenyl-1-ethyl-4-(2-morpholinoethyl)-2-pyrrolidinone hydrochloride
- Dopram
- Dothiepinhydrochloride,Anhydrous
- Mehr Synonyme anzeigen
- Doxapram hcl
- Doxapram hydrochloride anhydrous
- NSC 170958
- Stimulexin
- Unii-P5Ru6Uoq5Y
| Marke | Produktdaten | Reinheit | Preisklasse | Voraussichtliche Lieferung |
|---|---|---|---|---|
 | Doxapram hydrochloride REF: IN-DA0081M0CAS: 113-07-5 | - - - | Nachfragen | Di 10 Dez 24 |
 | Doxapram-d5 HCl REF: 4Z-D-142004CAS: 113-07-5 | - - - | Nachfragen | Di 10 Dez 24 |
 | Doxapram hydrochloride REF: 04-C13084100CAS: 113-07-5 | - - - | 161,00 € | Mi 11 Dez 24 |
 | Doxapram hydrochloride REF: 3D-FD61522CAS: 113-07-5 | Min. 95% | 136,00 €~723,00 € | Do 19 Dez 24 |
 | Doxapram Hydrochloride REF: TR-D552503CAS: | - - - | - - - | Ausgelaufenes produkt |

Ref: IN-DA0081M0
| Unbestimmte Größe | Nachfragen |

Doxapram-d5 HCl
Ref: 4Z-D-142004
| 10mg | Nachfragen | ||
| 25mg | Nachfragen | ||
| 50mg | Nachfragen | ||
| 100mg | Nachfragen |

Ref: 04-C13084100
| 10mg | 161,00 € |

Doxapram hydrochloride
Ref: 3D-FD61522
| 25mg | 136,00 € | ||
| 100mg | 303,00 € | ||
| 250mg | 457,00 € | ||
| 500mg | 723,00 € |

Ref: TR-D552503
| 100mg | Ausgelaufen | Anforderung von Informationen |