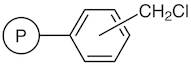
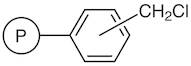
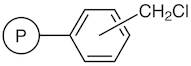
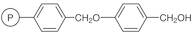
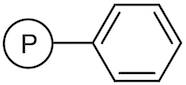
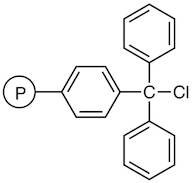
01-D-1250 - Wang Resin 200 400 Mesh 080 120 Mmolg
Sorry, no product with reference 01-D-1250has been found, but please check out the following similar products:
See more
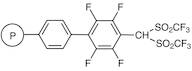
Bis(trifluoromethanesulfonyl)methyltetrafluorophenyl Polystyrene Resin cross-linked with 2% DVB (200-400mesh) (0.9-1.2mmol/g)
Color and Shape:
Orange to Brown to Dark red powder to crystal
Ref: 3B-B2292
| 100mg | 312.00 € |
Estimated delivery in United States, on Friday 26 Apr 2024

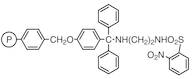
N-(4-Benzyloxytrityl)-N'-(2-nitrobenzenesulfonyl)-1,2-diaminoethane Resin cross-linked with 1% DVB (200-400mesh) (0.9-1.1mmol/g)
Color and Shape:
Solid
Ref: 3B-B2370
| 1g | 577.00 € |
Estimated delivery in United States, on Friday 26 Apr 2024

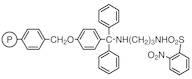
N-(4-Benzyloxytrityl)-N'-(2-nitrobenzenesulfonyl)-1,3-diaminopropane Resin cross-linked with 1% DVB (200-400mesh) (0.9-1.1mmol/g)
Color and Shape:
Solid
Ref: 3B-B2371
| 1g | 596.00 € |
Estimated delivery in United States, on Friday 26 Apr 2024

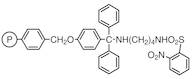
N-(4-Benzyloxytrityl)-N'-(2-nitrobenzenesulfonyl)-1,4-diaminobutane Resin cross-linked with 1% DVB (200-400mesh) (0.9-1.1mmol/g)
Color and Shape:
Solid
Ref: 3B-B2372
| 1g | 558.00 € |
Estimated delivery in United States, on Friday 26 Apr 2024

Chloromethyl Polystyrene Resin cross-linked with 2% DVB (200-400mesh) (2.0-2.7mmol/g)
CAS:55844-94-5
Color and Shape:
White to Light yellow powder to crystal
Ref: 3B-C1745
| 5g | 38.00 € |
Estimated delivery in United States, on Friday 26 Apr 2024

Chloromethyl Polystyrene Resin cross-linked with 1% DVB (100-200mesh) (2.0-3.0mmol/g)
CAS:55844-94-5
Color and Shape:
White to Light yellow powder to crystal
Ref: 3B-C1750
| 5g | 39.00 € | |
| 25g | 122.00 € |
Estimated delivery in United States, on Friday 26 Apr 2024

Chloromethyl Polystyrene Resin cross-linked with 2% DVB (100-200mesh) (0.8-1.2mmol/g)
CAS:55844-94-5
Color and Shape:
White to Orange to Green powder to crystal
Ref: 3B-C1751
| 5g | 39.00 € | |
| 25g | 99.00 € |
Estimated delivery in United States, on Friday 26 Apr 2024

4-(Hydroxymethyl)phenoxymethyl Polystyrene Resin cross-linked with 1% DVB (200-400mesh) (1.0-2.0mmol/g)
Color and Shape:
White to Light yellow to Light orange powder to crystal
Ref: 3B-H0987
| 5g | 100.00 € |
Estimated delivery in United States, on Friday 26 Apr 2024

Polystyrene Resin cross-linked with 1% DVB (200-400mesh)
CAS:9003-70-7
Color and Shape:
White to Almost white granule
Ref: 3B-P1379
| 25g | 78.00 € |
Estimated delivery in United States, on Friday 26 Apr 2024

Trityl Chloride Resin cross-linked with 1% DVB (200-400mesh) (2.0-2.5mmol/g)
Color and Shape:
White to Amber powder to crystal
Ref: 3B-T3432
| 1g | 84.00 € |
Estimated delivery in United States, on Friday 26 Apr 2024

Silver two part conductive adhesive, EXP. XX/XX Resin and Hardener
A conductive epoxy which bonds well to metal, ceramics, plastics, glass and graphite This Thermo …
Formula:
Limitedshelflifedaysaftershipment
Color and Shape:
Hardener: Colorless to pale yellow, Resin: Silver, Liquid
Ref: 02-012942
| 1g | 53.00 € | |
| 10g | 211.00 € |
Estimated delivery in United States, on Monday 29 Apr 2024

Amberlite™ IRN-150, ion exchange resin
Amberlite IRN-150 is used in water treatment and purification in primary side chemical volume control …
Formula:
StyreneDVB
Color and Shape:
Yellow to brown, Spherical particles
Ref: 02-040992
| 2kg | 185.00 € | |
| 10kg | 776.00 € | |
| 500g | 62.00 € |
Estimated delivery in United States, on Monday 29 Apr 2024

Graphite plate, resin impregnated, 6.35mm (0.25in) thick
Graphite is basically used for casting and polishing purposes, moulding & carbon addition in steel …
Color and Shape:
Solid
Ref: 02-041831
| 100x100mm | To inquire |
Estimated delivery in United States, on Monday 29 Apr 2024

Amberlite™ IRA-200C(Na), ion exchange resin
Amberlite™ IRA-200C(Na), ion exchange resin is used for makeup demineralization, hot process softeners and sodium …
Color and Shape:
Spherical particle, Pale yellow to pale brown
Ref: 02-042252
| 1kg | 64.00 € | |
| 5kg | 213.00 € | |
| 250g | 34.00 € |
Estimated delivery in United States, on Monday 29 Apr 2024

Amberlite™ IRA-67, ion exchange resin
CAS:80747-90-6
Amberlite™ IRA-67 is used for de-acidification and desalination of water and for isolation of acidic …
Color and Shape:
Solid Beads
Ref: 02-042253
| 1kg | 144.00 € | |
| 5kg | 370.00 € | |
| 250g | 48.00 € |
Estimated delivery in United States, on Monday 29 Apr 2024

Amberlite™ IRA-958(Cl), ion exchange resin
Removal of color, organics, and cyanideAmberlite™ IRA-958(Cl), ion exchange resin is used as an organic …
Color and Shape:
White to pale cream, Spherical beads
Ref: 02-042702
| 1kg | 172.00 € | |
| 5kg | 524.00 € | |
| 250g | 72.00 € |
Estimated delivery in United States, on Monday 29 Apr 2024

Amberlyst™ 15(H), wet, ion exchange resin
CAS:39389-20-3
Amberlyst™ 15(H) is a strongly acidic cation exchange resin used as a heterogeneous acid catalysis …
Formula:
C18H18O3S
Color and Shape:
Opaque, Beads
Molecular weight:
314.40
Ref: 02-044079
| 1kg | 123.00 € | |
| 5kg | 380.00 € | |
| 250g | 39.00 € |
Estimated delivery in United States, on Monday 29 Apr 2024

Amberlyst™ 15(H), ion exchange resin
CAS:39389-20-3
Amberlyst™ 15(H) is a strongly acidic cation exchange resin used as a heterogeneous acid catalysis …
Formula:
C18H18O3S
Color and Shape:
Light Brown Solid Beads
Molecular weight:
314.40
Ref: 02-089079
| 1kg | 259.00 € | |
| 50g | 29.00 € | |
| 250g | 89.00 € |
Estimated delivery in United States, on Monday 29 Apr 2024

Amberlyst™ A-26(OH), ion exchange resin
CAS:39339-85-0
Amberlyst™ A-26(OH) is widely utilized to catalyze reactions such as aldol condensation and carbonylation. It …
Color and Shape:
Beads, Moist pink or pale brown
Ref: 02-A17361
| 1kg | 232.00 € | |
| 250g | 80.00 € |
Estimated delivery in United States, on Monday 29 Apr 2024

Amberlite IRA-410(Cl), ion exchange resin
CAS:9002-26-0
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. …
Color and Shape:
Beads, Pale yellow to pale orange
Ref: 02-A17734
| 100g | 31.00 € | |
| 500g | 77.00 € | |
| 2500g | 297.00 € |
Estimated delivery in United States, on Monday 29 Apr 2024