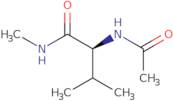
Ac-Val-NHMe
CAS: 19701-84-9
Ref. 3D-FA111816
| 1g | To inquire | |
| 2g | To inquire | |
| 5g | To inquire | |
| 10g | To inquire | |
| 500mg | To inquire |
Product Information
- (2S)-2-(Acetylamino)-N,3-dimethylbutanamide
- (2S)-2-Acetamido-N,3-dimethylbutanamide
- 2-Acetamido-N,3-dimethyl-<span class="text-smallcaps">L</span>-butyramide
- Butanamide, 2-(acetylamino)-N,3-dimethyl-, (2S)-
- Butanamide, 2-(acetylamino)-N,3-dimethyl-, (S)-
- Butyramide, 2-acetamido-N,3-dimethyl-, <span class="text-smallcaps">L</span>-
- N-Acetyl-<span class="text-smallcaps">L</span>-valine N-methylamide
- N-Acetyl-<span class="text-smallcaps">L</span>-valine methylamide
- N-Acetyl-N′-methyl-<span class="text-smallcaps">L</span>-valinamide
- N-Acetyl-N′-methylvalinamide
- See more synonyms
- N~2~-acetyl-N-methyl-L-valinamide
Ac-Val-NHMe is a non-polar, hydrophobic molecule that participates in non-covalent interactions with other molecules. The main function of Ac-Val-NHMe is the formation of hydrogen bonds and van der Waals interactions with other hydrophobic molecules. It has been shown experimentally that Ac-Val-NHMe interacts with norvaline to form a dimer. This dimer can be rationalized by the theory of rationalized molecular orbital (MO) theory and functional theory. The dihedral angle for this interaction is 100°, which is chiral. However, at low temperatures, it may also exist as a racemic mixture due to the lack of sufficient energy to overcome the entropy barrier.
Chemical properties
Technical inquiry about: 3D-FA111816 Ac-Val-NHMe
If you want to request a quotation or place an order, please instead add the desired products to your cart and then request a quotation or order from the cart. It is faster, cheaper, and you will be able to benefit from the available discounts and other advantages.