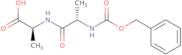
Product Information
- Z-L-alanyl-L-alanine
- <span class="text-smallcaps">L</smallcap>-Alanine, N-[(phenylmethoxy)carbonyl]-<smallcap>L</span>-alanyl-
- <span class="text-smallcaps">L</smallcap>-Alanine, N-[N-[(phenylmethoxy)carbonyl]-<smallcap>L</span>-alanyl]-
- Alanine, N-(N-carboxy-<span class="text-smallcaps">L</smallcap>-alanyl)-, N-benzyl ester, <smallcap>L</span>-
- Alanine, N-(N-carboxyalanyl)-, N-benzyl ester
- Benzyloxycarbonyl-<span class="text-smallcaps">L</smallcap>-alanyl-<smallcap>L</span>-alanine
- N-(Benzyloxycarbonyl)-<span class="text-smallcaps">L</smallcap>-alanyl-<smallcap>L</span>-alanine
- N-(Benzyloxycarbonyl)alanylalanine
- N-[(Phenylmethoxy)carbonyl]-<span class="text-smallcaps">L</smallcap>-alanyl-<smallcap>L</span>-alanine
- N-[(benzyloxy)carbonyl]alanylalanine
- See more synonyms
Z-Ala-Ala-OH is an amide that has been shown to inhibit the activity of serine proteases, such as elastase and cathepsin G. It has been used to study the biological function of these enzymes in mammalian tissue. Z-Ala-Ala-OH has been shown to inhibit the activity of a number of other proteases, including trypsin, chymotrypsin, and plasmin. The phosphate group on this molecule may be hydrolyzed by phosphatases, which are found in many cells. This hydrolysis may result in the release of acetic acid, acetaldehyde, and ammonia. Z-Ala-Ala-OH is synthesized from cyclobutanone by an acylation reaction with a carboxylic acid or an amine.
Chemical properties
Technical inquiry about: 3D-FA47260 Z-Ala-Ala-OH
If you want to request a quotation or place an order, please instead add the desired products to your cart and then request a quotation or order from the cart. It is faster, cheaper, and you will be able to benefit from the available discounts and other advantages.