Product Information
- Xeloda, Capecitibine, Capiibine, RO 09-1978
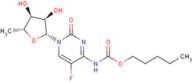
- 1-(5-deoxypentofuranosyl)-5-fluoro-4-{[(pentyloxy)carbonyl]amino}pyrimidin-2(1H)-one
- 5-Deoxy-5-fluoro-N-[(pentyloxy)carbonyl]cytidine
- 5′-Deoxy-5-fluoro-N<sup>4</sup>-(pentyloxycarbonyl)cytidine
- Capacetrine
- Capecitabine Accord
- Capecitabine Medac
- Capecitabine SUN
- Capecitabine Teva
- Capecitibine
- See more synonyms
- Capecytabine
- Capiibine
- Captabin
- Carbamic acid, [1-(5-deoxy-β-<span class="text-smallcaps">D</span>-ribofuranosyl)-5-fluoro-1,2-dihydro-2-oxo-4-pyrimidinyl]-, pentyl ester
- Cytidine, 5′-deoxy-5-fluoro-N-[(pentyloxy)carbonyl]-
- Ecansya
- N4-[(pentyloxy)carbonyl]-5'-deoxy-5-fluorocytidine
- N<sup>4</sup>-Pentyloxycarbonyl-5′-deoxy-5-fluorocytidine
- Pentyl [1-(3,4-dihydroxy-5-methyl-oxolan-2-yl)-5-fluoro-2-oxo-pyrimidin-4-yl]aminoformate
- R-340
- Ro 09-1978/000
- Ro-09-1978
- Xeloda
- Xeltabin
Capecitabine (Capecitibine) is a fluoropyrimidine carbamate belonging to the class of antineoplastic agents called antimetabolites. As a prodrug, capecitabine is selectively activated by tumor cells to its cytotoxic moiety, 5-fluorouracil (5-FU); subsequently, 5-FU is metabolized to two active metabolites, 5-fluoro-2-deoxyuridine monophosphate (FdUMP) and 5-fluorouridine triphosphate (FUTP) by both tumor cells and normal cells. FdUMP inhibits DNA synthesis and cell division by reducing normal thymidine production, while FUTP inhibits RNA and protein synthesis by competing with uridine triphosphate for incorporation into the RNA strand.
Chemical properties
Technical inquiry about: TM-T1408 Capecitabine
If you want to request a quotation or place an order, please instead add the desired products to your cart and then request a quotation or order from the cart. It is faster, cheaper, and you will be able to benefit from the available discounts and other advantages.