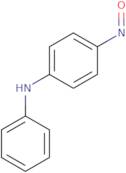
4-Nitrosodiphenylamine
CAS: 156-10-5
Ref. 3D-AAA15610
| 1g | 63.00 € | ||
| 2g | 97.00 € | ||
| 5g | 138.00 € | ||
| 10g | 194.00 € | ||
| 25g | 348.00 € |
Product Information
- 4-nitroso-N-phenylaniline
- (4-Nitroso-phenyl)-phenyl-amine
- 4-Nitroso-N-phenylbenzenamine
- Amine, diphenyl, 4-nitroso-
- Benzenamine, 4-nitroso-N-phenyl-
- Brn 1952651
- Ccris 465
- Diphenylamine, 4-nitroso-
- Hsdb 2876
- N-Phenyl-4-nitrosoaniline
- See more synonyms
- N-Phenyl-p-nitrosoaniline
- N-Phenyl-para-nitrosoaniline
- N-nitroso-N-phenylaniline
- Naugard TKB
- Nci-C02244
- Nitrous diphenylamide
- Nsc 5041
- TKB
- p-Nitroso-N-phenylaniline
- p-Nitrosodifenylamin
- p-Nitrosodifenylamin [Czech]
- p-Nitrosodiphenylamine
- p-Phenylaminonitrosobenzene
- para-Nitroso-N-phenylaniline
4-Nitrosodiphenylamine (4NDS) belongs to the class of nitrosamines and is an aliphatic hydrocarbon. It is a potential carcinogenic with an optimum concentration of 0.1ppm, but has not been shown to be carcinogenic at dietary concentrations. 4NDS reacts with sodium hydroxide solution and hydroxide solution to form a cationic surfactant that can be used as a detergent in cleaning products, which may lead to skin irritation. The mechanism of the reaction is through an acylation reaction involving the formation of an amide bond between the nitrogen atom in 4NDS and the carbon atom in a carboxylic acid. The activated 4NDS molecule then reacts with ozone, forming nitric oxide, which reacts with hydrogen peroxide to form water and oxygen gas, thereby destroying bacteria on surfaces.
Chemical properties
Technical inquiry about: 3D-AAA15610 4-Nitrosodiphenylamine
If you want to request a quotation or place an order, please instead add the desired products to your cart and then request a quotation or order from the cart. It is faster, cheaper, and you will be able to benefit from the available discounts and other advantages.