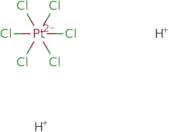
Chloroplatinic acid hydrate
CAS: 26023-84-7
Ref. 3D-FC158320
| 1g | To inquire | |
| 2g | To inquire | |
| 5g | To inquire | |
| 250mg | To inquire | |
| 500mg | To inquire |
Product Information
- Dihydrogen hexachloroplatinate hexahydrate
- Dihydrogen hexachloroplatinate(2-) hydrate
- Dihydrogen hexachloroplatinate(IV) hexahydrate
- Hexachloroplatinate dihydrogen hydrate
- Hexachloroplatinic acid hydrate
- Hexachloroplatinic(IV) acid 6H2O
- Hydrogen Hexachloroplatinate(2-) Hydrate (2:1:6)
- Hydrogen hexachloroplatinate(IV) hexahydrate
- Hydrogen hexachloroplatinate(IV) hydrate
- Platinate(2-), hexachloro-, dihydrogen, hexahydrate
- See more synonyms
- Platinate(2-), hexachloro-, dihydrogen, hexahydrate, (OC-6-11)-
- Platinate(2-), hexachloro-, dihydrogen, hydrate
- Platinate(2-), hexachloro-, dihydrogen, hydrate, (OC-6-11)-
- Platinate(2-), hexachloro-, hydrogen, hydrate (1:2:?), (OC-6-11)-
- Platinum chloric acid dihydrochloride hexahydrate
Chloroplatinic acid hydrate is a chemical compound that belongs to the group of platinum compounds. It is an effective catalyst for a variety of reactions, including photocatalytic activity, cross-linking of beta-lactamase inhibitors with phosphotungstic acid, and electrochemical studies. The optimum concentration for chloroplatinic acid hydrate is 15 mg/mL in hydrochloric acid solution. The concentration in urine samples can be determined by gravimetric analysis or by measuring the amount of platinum electrodes in the solution before and after reaction with chloroplatinic acid hydrate. Chloroplatinic acid hydrate can be synthesized by reacting hydrogen chloride with platinum metal under an inert atmosphere.
Chemical properties
Technical inquiry about: 3D-FC158320 Chloroplatinic acid hydrate
If you want to request a quotation or place an order, please instead add the desired products to your cart and then request a quotation or order from the cart. It is faster, cheaper, and you will be able to benefit from the available discounts and other advantages.