Product Information
- Potassium Oxido(Dioxo)Vanadium
- Potassium m-vanadate
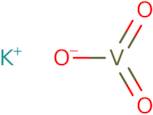
- Potassium metavanadate (KVO<sub>3</sub>)
- Potassium metavanadate, 99.9% (metals basis)
- Potassium trioxovanadate
- Potassium vanadate (KVO<sub>3</sub>)
- Potassium vanadate(V) (KVO<sub>3</sub>)
- Potassium vanadium trioxide
- Vanadate (VO<sub>3</sub><sup>1-</sup>), potassium
- Vanadate (VO<sub>3</sub><sup>1-</sup>), potassium (1:1)
- See more synonyms
- Vanadate, potassium
- Vanadic acid (HVO<sub>3</sub>), potassium salt
Potassium metavanadate is a compound that has been used to remove uranium from water. It binds to uranium by hydrogen bonding interactions and forms a soluble complex that can be removed from the water. The removal of uranium with potassium metavanadate is limited by the matrix effect, which refers to the ability of the sorbent to bind other ions in solution. The reaction mechanism for potassium metavanadate involves oxidation of vanadium (V) ions to form vanadates (VO). This process is reversible and can be reversed by reducing vanadium ions with hydrogen gas or copper chloride. The phase transition temperature for this reaction is 230°C, so potassium metavanadate will not work well at higher temperatures. Potassium metavanadate is an example of group 2 metals because it contains two metal atoms: one of which is a transition metal (vanadium) and the other is alkali metal (potassium). Its coordination geometry is octahedral.
Chemical properties
Technical inquiry about: 3D-FP170862 Potassium metavanadate
If you want to request a quotation or place an order, please instead add the desired products to your cart and then request a quotation or order from the cart. It is faster, cheaper, and you will be able to benefit from the available discounts and other advantages.