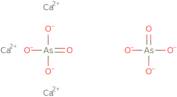
Arsenic acid (H3AsO4),calcium salt (2:3)
CAS: 7778-44-1
Ref. 3D-HAA77844
| 25g | To inquire | ||
| 50g | To inquire | ||
| 100g | To inquire | ||
| 250g | To inquire | ||
| 500g | To inquire |
Product Information
- Arsenic acid (H3AsO4), calcium salt (2:3)
- Arsenic acid (H<sub>3</sub>AsO<sub>4</sub>), calcium salt (2:3)
- Arsenic acid, calcium salt (2:3)
- Calcium Arsenate (3:2)
- Calcium arsenate (Ca<sub>3</sub>(AsO<sub>4</sub>)<sub>2</sub>)
- Calcium arsenate [2AsH3O4.2Ca]
- Calcium arsenate, 99% (metals basis)
- Chip-Cal Granular
- Chip-cal
- Cucumber dust
- See more synonyms
- Pencal
- Security
- Spra-cal
- Tricalcium Arsenate
- Tricalcium orthoarsenate
- Turf-Cal
Arsenic Acid is a compound made up of arsenic and oxygen. It is acidic and has an activation energy of 15.6 kcal/mol, a thermodynamic data of -121.5 kJ/mol, and a neutral ph. Arsenic acid reacts with sodium hydroxide solution to form sodium arsenite and hydrogen gas. This reaction is exothermic, meaning that it releases heat. The dry weight of arsenic acid is 1.07 grams per mole and the metal hydroxides are calcium (1:1) and magnesium (2:3). X-ray diffraction data shows that calcium has low energy peaks at 13 degrees, 16 degrees, 18 degrees, and 20 degrees. A membrane system can be used to remove arsenic from wastewater treatment by using an anion exchange resin in conjunction with an organic co-solvent such as methanol or acetonitrile.
Chemical properties
Technical inquiry about: 3D-HAA77844 Arsenic acid (H3AsO4),calcium salt (2:3)
If you want to request a quotation or place an order, please instead add the desired products to your cart and then request a quotation or order from the cart. It is faster, cheaper, and you will be able to benefit from the available discounts and other advantages.