Product Information
The functional theory is based on the assumption that each chemical reaction has an associated set of molecular orbitals. When the orbitals are occupied by electrons, they can interact with other molecules to form a new set of orbitals. The stepwise theory proposes that reactions take place in a series of small steps, each involving only one chemical species. In proton transfer reactions, the proton is transferred from one molecule to another by means of a hydrogen bond. This process can be described as an electron-proton coupling mechanism, which is found in many organic compounds. Hydroxyl groups are intramolecular hydrogen bonds between hydroxyl and oxygen atoms in neighboring molecules. These bonds are formed through hydrogen bonding and often break down when exposed to heat or light. Hydrogen bonds between molecules can be classified as either intermolecular or intramolecular depending on whether they occur between molecules or within a single molecule. Intermolecular hydrogen bonds occur between separate molecules whereas intramolecular
Chemical properties
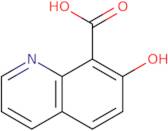
Technical inquiry about: 3D-WVB29853 7-Hydroxyquinoline-8-carboxylic acid
If you want to request a quotation or place an order, please instead add the desired products to your cart and then request a quotation or order from the cart. It is faster, cheaper, and you will be able to benefit from the available discounts and other advantages.